 Stent graft design used to treat Thoracic Aortic Aneurysms
Stent graft design used to treat Thoracic Aortic AneurysmsMedtronic: Advancing Treatment and Improving Lives
Medtronic is the global leader in medical technology. The Endovascular division designs and manufactures devices that treat cardiovascular disease such as Thoracic Aortic Aneurysms. Medtronic Endovascular commits unwaveringly to improving lives with patient outreach, educational programs that raise awareness of cardiovascular disease, and the continuing pursuit of new treatment options. Medtronic Endovascular is proud to have helped physicians treat over 200,000 patients worldwide.
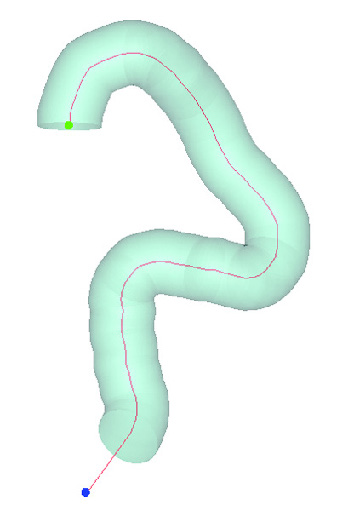 Aortic model generated from centerline and geometric parameters
Aortic model generated from centerline and geometric parametersNew Ways To Design Stent Test Models
When testing grafts used to treat Thoracic Aortic Aneurysms, Medtronic’s goal is to develop models that help to accurately mimic in-vivo device performance. Due to the critical role these grafts play in a patient’s well-being, a new method of designing test models was developed. By incorporating statistical analysis with the development of benchtop test models, Medtronic is able to ensure that their devices will perform under challenging conditions.
Using the Mimics Innovation Suite, Medtronic developed a method of obtaining geometric parameters from actual patient data to define the in-vivo use conditions. Patient CT data from the field was collected and delivered to the research and development team. Using Materialise’s Mimics software, the team segmented the 3D aortic model from the datasets. A centerline was automatically calculated in Mimics to fit the aortic model. To describe challenge-use conditions, the centerline was morphed to fit the 95th percentile value for each geometric parameter. Using this hybrid method of combining actual patient data and statistically assessed geometric parameters, the vascular models are able to be adapted to fit any requirements for testing purposes.
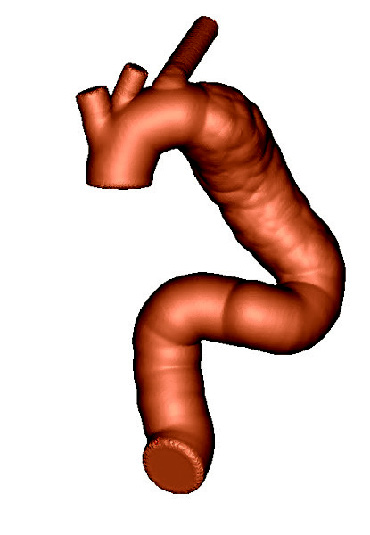 Patient-based aortic model
Patient-based aortic modelAfter forming the hybrid centerline, Materialise’s 3-matic software was used to design a thin walled patient-based aortic model. Supports and standard test-fittings were also designed into the device before using 3D Printing technology to print a physical benchtop test model.
The model was then fitted into the benchtop test apparatus with cycling fluid to evaluate and quantify several performance metrics of the stent graft and its delivery system.
Medtronic’s method for designing a patient-based benchtop test apparatus can be summarized in five steps:
- Measure geometric parameters
- Calculate centerline
- Adapt centerline to fit statistical models
- Design test apparatus
- Print model with 3D Printing for medical technology
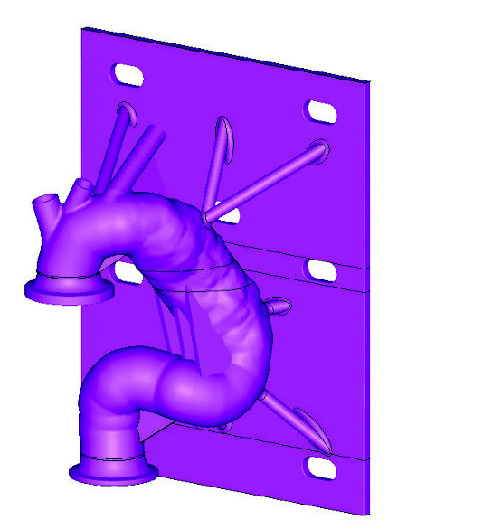 Patient-based aortic model
Patient-based aortic modelPromising Results Achieved with the Mimics Innovation Suite
The use of the Mimics Innovation Suite to quantify anatomical geometry and generate a set of standard patient-based models has created an unsurpassed standard for benchtop testing at Medtronic. “Mimics allows us to quantify edge of failure conditions and 3-matic can incorporate those performance limits into next generation device development test models,” says Srinivasan Varahoor. These models can help systematically pin-point conditions for possible device failure during testing, and thereby, result in more robust designs. This approach is applicable to the testing and development for any vascular device system in the future.





